 Taken in these types of determinations however. Additional distances canīe obtained from other distances between atoms. Therefore theĪtomic radius of chlorine is 0.994 Å. Nuclei is the sum of two chlorine atomic radii. To get the atomic radius we assume the distance between the two The two chlorine atoms in Cl 2 is known to be 1.988 Å. It is difficult toĭefine a sharp boundary for distance between the electrons in any Very broad region for finding the electron. Our quantum mechanical description of an atom suggests a The immediate question is what is an atomic The first property to explore isĪtomic radius. The Quantum Mechanical model of theĪtom can 'tested' by looking at the experimental data of atomic The strength of any model is in it ability to explainĮxperimental observations. It does not store any personal data.The first property to explore is atomic radius. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary".  
The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. The two atomic radius trends we discussed above are true for the majority of the periodic table of elements. The potassium atom has an extra electron shell compared to the sodium atom, which means its valence electrons are further from the nucleus, giving potassium a larger atomic radius. Why does potassium have a larger atomic radius than sodium? The six noble gases, in group 18 of the periodic table, are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). However, there are a few exceptions to these trends. An atomic radius is one-half the distance between the nuclei of two atoms.Īre there any exceptions to the atomic radius trend? A radius is the distance between the center of an object and its outer edge. Which is true about the radius of an atom?Īn atom is a basic unit of a chemical element, such as hydrogen, helium, potassium, etc. When an additional shell is added, those new electrons are farther from the atom’s nucleus, which increases atomic radius. The first atomic radius periodic trend is that atomic radii increase as you move downwards in a group. Why does atomic radius increase as you move down a group? The atomic radius of atoms generally increases from top to bottom within a group. The atomic radius of atoms generally decreases from left to right across a period. 
How do atomic radii vary in a group and period? The growing size is due to the increasing number of filled electron shells as you move down the periodic table. The bigger the atomic number, the larger the atom’s radius. 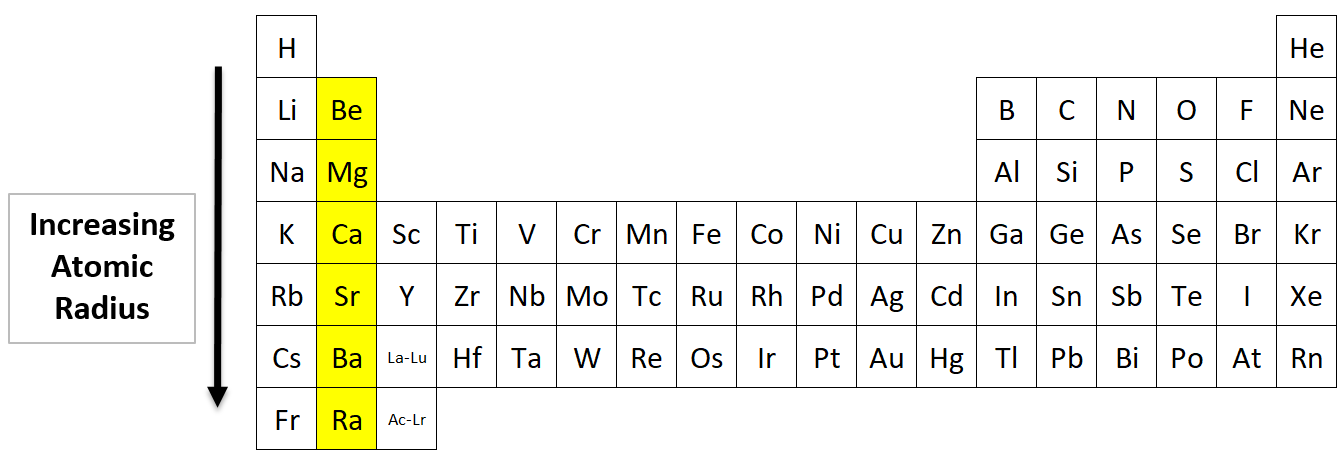
What affects the atomic radius?Īs the atomic number of an element increases, so does the size of its nucleus and the number of electrons around it. As we move from left to right across a period on a periodic table, the number of protons in the nucleus of the atom increases. Farther the last shell from nucleus, greater is the atom’s size. The atomic size is determined by how far the last shell of electrons is away from the nucleus. Why does the atomic radius increase from right to left? As a result, the atomic radius decreases. The valence electrons are held closer towards the nucleus of the atom. This is because, within a period or family of elements, all electrons are added to the same shell. Why does the atomic radius decrease across a period?Ītomic size gradually decreases from left to right across a period of elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
